Health Library
Mazdutide vs Tirzepatide: How These Weight Loss Drugs Compare
February 21, 2026
Question on this topic? Get an instant answer from August.
Mazdutide and tirzepatide are both injectable medications that target gut hormones to help with weight loss and blood sugar control. The key difference is in how they work. Tirzepatide activates GLP 1 and GIP receptors, while mazdutide activates GLP 1 and glucagon receptors. This distinction affects how each drug influences metabolism, appetite, and energy expenditure.
Both have shown strong results in clinical trials, but they are at very different stages of availability. Here a closer look at how they compare.
How Does Each Drug Work?
Understanding mechanism helps explain why these drugs produce different effects.
Tirzepatide (sold as Mounjaro for type 2 diabetes and Zepbound for weight management) is a dual GIP/GLP 1 receptor agonist. GLP 1 slows gastric emptying, reduces appetite, and helps regulate blood sugar. GIP (glucose dependent insulinotropic polypeptide) enhances insulin secretion and may also influence fat metabolism. The combination of these two pathways what gives tirzepatide its powerful effect on both weight and blood sugar.
Mazdutide takes a different dual approach. It activates GLP 1 receptors and glucagon receptors. The GLP 1 component reduces appetite and slows digestion, similar to tirzepatide. The glucagon component adds something tirzepatide does not directly offer: it may increase energy expenditure. Glucagon stimulates liver to release stored energy and may promote fat burning, which could lead to greater fat loss beyond what appetite suppression alone can achieve.
How Much Weight Loss Does Each Drug Produce?
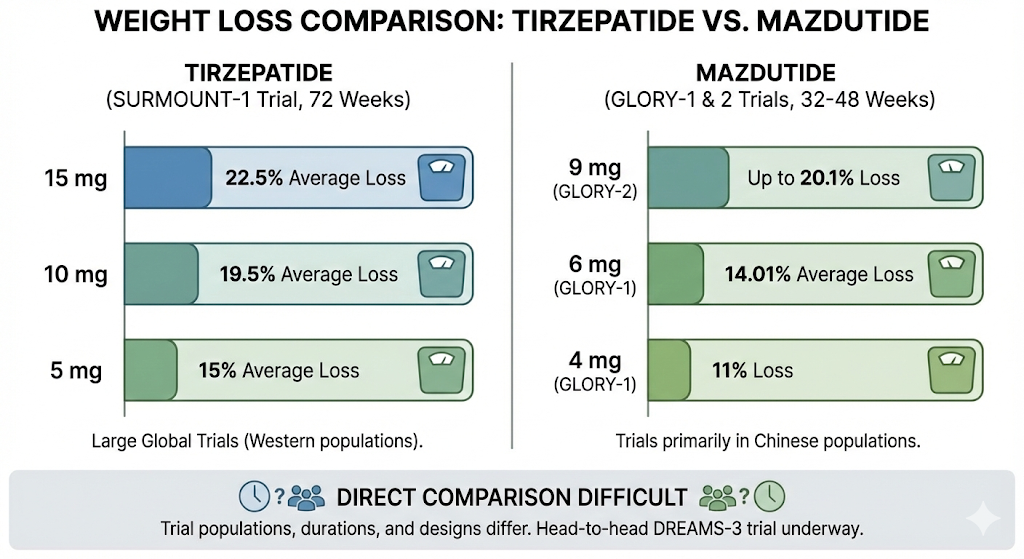
Tirzepatide has strong, well established weight loss data from large global trials. In SURMOUNT 1 trial, participants without diabetes who received highest dose (15 mg) lost an average of 22.5% of their body weight over 72 weeks. Lower doses (5 mg and 10 mg) produced weight losses of around 15% and 19.5% respectively.
Mazdutide's data comes primarily from trials conducted in Chinese populations. In GLORY 1 phase 3 trial published in New England Journal of Medicine, adults with overweight or obesity who received mazdutide 6 mg lost an average of 14.01% of body weight over 48 weeks. The 4 mg dose produced about 11% weight loss. More recently, GLORY 2 trial testing a higher 9 mg dose reported weight loss of up to 20.1% in adults with obesity.
A direct comparison is difficult because trial populations, durations, and designs differ. Tirzepatide's largest trials were conducted in Western populations over longer durations (72 weeks), while mazdutide's phase 3 data comes from shorter trials (32 to 48 weeks) in Chinese adults with different BMI thresholds. A head to head trial (DREAMS 3) comparing mazdutide directly against semaglutide in patients with type 2 diabetes and obesity is underway, which should offer more useful comparative data.
What About Blood Sugar Control?
Both drugs have significant effects on blood sugar, making them relevant for people with type 2 diabetes.
Tirzepatide has been extensively studied for diabetes management. In SURPASS trials, tirzepatide at 15 mg reduced HbA1c (a marker of average blood sugar over three months) by about 2.3 percentage points. Many participants achieved HbA1c levels below 5.7%, which is considered normal, non diabetic range.
Mazdutide has also shown meaningful HbA1c reductions in people with type 2 diabetes. In its DREAMS trials, mazdutide improved blood sugar levels alongside weight loss. The glucagon receptor activation in mazdutide could theoretically raise blood sugar, since glucagon promotes glucose release from liver. But in practice, GLP 1 component counterbalances this, and overall effect on blood sugar has been favorable in trials conducted so far. If you are interested in how dosing transitions between similar medications work, this guide on semaglutide to tirzepatide conversion explains process clearly.
What Are Side Effects?
The side effect profiles of both drugs are dominated by gastrointestinal symptoms. This expected with any medication that works through GLP 1 pathway.
For tirzepatide, most commonly reported side effects include nausea, diarrhea, vomiting, constipation, and decreased appetite. These symptoms are usually most noticeable during dose escalation period and tend to improve as body adjusts. Sulfur burps are another frequently reported complaint among tirzepatide users. If that something you have experienced, this article on why Zepbound causes sulfur burps explains what is behind it and how to manage it.
Mazdutide shows a similar gastrointestinal side effect pattern. In GLORY 1 trial, most common adverse events were gastrointestinal and mostly mild to moderate. Diarrhea, nausea, and upper respiratory tract infections were among most frequently reported. Importantly, discontinuation rate due to side effects was very low, just 0.5% to 1.5% in mazdutide groups.
One area to watch with mazdutide is heart rate. Like other GLP 1 based therapies, mazdutide has been associated with a slight increase in resting heart rate in clinical trials. This consistent with what has been seen with tirzepatide and semaglutide as well, but it remains something researchers are monitoring closely in longer term studies.
Which Drug Might Be More Effective Long Term?
It is too early to say definitively. Tirzepatide has a larger body of evidence from diverse, global populations and longer duration trials. Its weight loss results at 72 weeks are among strongest seen for any anti obesity medication.
Mazdutide's glucagon receptor activity is a unique feature that could potentially offer advantages in fat loss and metabolic improvement that go beyond appetite suppression. Early data promising, with up to 20.1% weight loss at 9 mg dose and strong improvements in liver fat and cardiometabolic markers. But longer trials with more diverse populations are needed before firm conclusions can be drawn.
Bottom Line
Tirzepatide and mazdutide are both powerful dual receptor agonists, but they target different hormone pathways. Tirzepatide combines GLP 1 and GIP activity and widely available with extensive clinical evidence. Mazdutide combines GLP 1 and glucagon activity, offers a potentially distinct metabolic profile, and showing impressive early results, but still in earlier stages of global development. For people already using tirzepatide, question whether mazdutide's glucagon component will offer meaningful additional benefits. For those in China, mazdutide already an approved option. The coming years of head to head trials and real world use will bring much clearer answers.
Health Companion
trusted by
6Mpeople
Get clear medical guidance
on symptoms, medications, and lab reports.